By manoj.kumar · January 19, 2026
You feel the impact of healthcare data silos every day. Your teams toggle between EMRs, claims platforms, lab systems, imaging, patient engagement tools, and population health dashboards. Each system holds a fragment of the patient story. Few of them talk to each other cleanly.
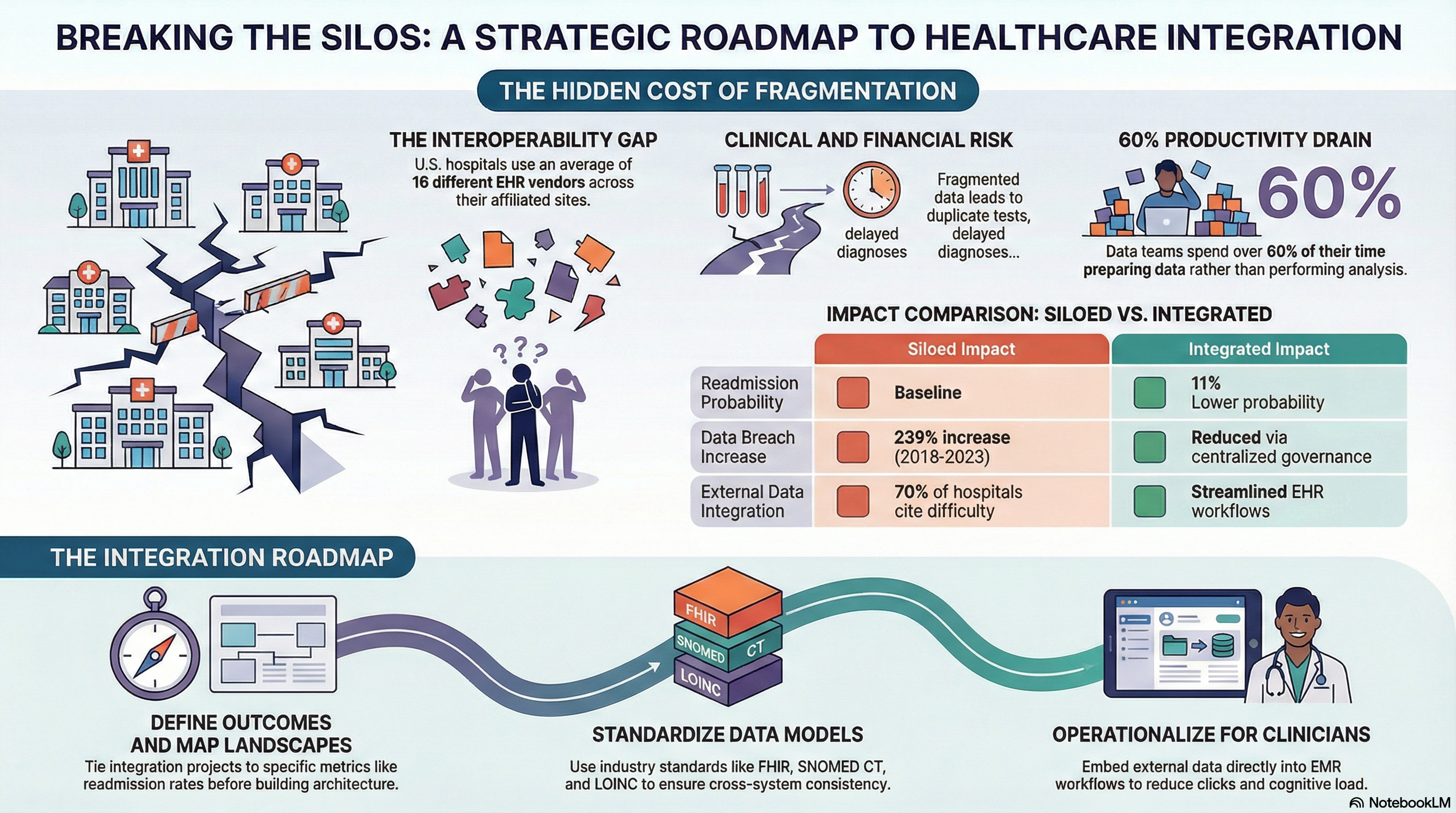
The result is slower decisions, higher risk, and frustrated clinicians who do not trust the data they see. One study found that U.S. hospitals use an average of 16 different EHR vendors across affiliated sites. At the same time, only 62 percent of hospitals routinely receive summary of care records electronically from outside providers. That gap shows how entrenched healthcare data silos are.
You do not solve this with another point solution. You solve it with a clear healthcare data strategy, a realistic integration roadmap, and disciplined governance. This guide breaks down why silos persist, where they put your health system at risk, and the practical steps you can take to achieve reliable health system integration.
Why Data Silos Exist
Fragmented technology decisions over many years
Most large health systems grew through mergers and acquisitions. Each acquired hospital or clinic brought its own EMR, revenue cycle system, lab platform, and imaging tools. During integration, clinical and financial operations often took priority. Legacy systems stayed in place, and interfaces became the quick fix.
Over a decade, those decisions stacked up. You now support parallel EMRs, duplicate registries, and separate data warehouses for service lines. The cost and risk of a single EMR migration feel high, so you live with partial EMR data consolidation through a patchwork of interfaces and batch feeds.
Department‑driven purchasing
Clinical departments often select niche applications that solve an immediate problem. Oncology picks one registry. Cardiology picks another. Population health teams bring in yet another platform.
Without a shared architectural review, these systems arrive with proprietary formats and unique APIs. Each one becomes another isolated store of data that must be integrated later, usually under time pressure.
Limitations of legacy integration approaches
Many integration teams still rely on point‑to‑point HL7 interfaces for every new connection. Each new app triggers a custom feed for ADT, orders, results, and charges. Over time, you end up with hundreds of brittle connections.
Changes in one system break several interfaces. Interface teams then spend their time keeping lights on rather than advancing hospital interoperability. The integration pattern itself reinforces healthcare data silos.
Vendor lock‑in and proprietary formats
Some platforms limit API access or charge high fees for integration. Others use proprietary data models that do not align with FHIR resources or standard terminologies. That friction slows down data sharing and encourages local workarounds, like shadow databases and spreadsheets.
Over time, those workarounds become unofficial sources of truth. You end up with parallel datasets for the same patients and encounters, each with different levels of quality.
Risks of Fragmentation
Clinical risk and incomplete patient context
When clinicians lack a full view of medications, allergies, and prior imaging, risk increases. The Office of the National Coordinator reported that over 70 percent of hospitals cite difficulty integrating external data into EHR workflows as a barrier to effective exchange.
Fragmented data leads to duplicate tests, delayed diagnoses, and avoidable adverse events. It also adds cognitive load, because clinicians must search across multiple systems for answers during time‑sensitive encounters.
Financial leakage and operational friction
Without integrated data, you struggle to see true cost and margin by service line, site, or population. Charge capture gaps hide in mismatched encounter IDs. Denials management teams piece together information across EMR, clearinghouse, and payer portals.
Health systems that achieve high interoperability performance report lower readmission penalties and higher quality incentive revenue. A study in Health Affairs found that hospitals engaged in more extensive electronic exchange hhadal 11 percent lower probability of 30-day readmissions for Medicare beneficiaries.
Regulatory and privacy exposure
Siloed data environments often include shadow copies, local extracts, and unmanaged data stores. Each one introduces security risk and complicates HIPAA compliance. The U.S. Department of Health and Human Services reported a 239 percent increase in large healthcare breaches between 2018 and 2023.
When you lack a consistent view of where PHI lives, you struggle to respond to right‑of‑access requests, right‑to‑amend requests, and breach investigations. Fragmentation turns every audit into a manual data hunt.
Blocked analytics and digital innovation
Your data science and analytics teams spend most of their time acquiring, cleaning, and reconciling data. That slows down everything from SDOH analytics to value‑based care performance reporting.
One survey found that data teams spend over 60 percent of their time on preparation work instead of analysis and model development. Healthcare data silos turn digital innovation roadmaps into integration backlogs.
Step‑by‑Step Integration
1. Clarify the outcomes before the architecture
Start with a specific set of outcomes that your integration work must support. Examples include:
• Reduce avoidable readmissions for high‑risk populations
• Improve referral completion and closed‑loop communication
• Support a new value‑based contract with complete quality reporting
• Enable longitudinal patient views in the EMR
Tie each outcome to clear metrics, such as readmission rate, referral completion rate, or time to insights for new reports. Your healthcare data strategy should connect every integration project to one of these outcomes.
2. Map your current data landscape
Build an inventory of:
• All clinical and operational systems that store PHI
• Existing interfaces, feeds, and APIs for each system
• Data domains: patient, provider, encounter, orders, results, claims
• Reference data sources: codes, providers, locations
Use this inventory to visualize clusters of healthcare data silos. Look for areas where the same data domain appears in multiple systems, such as patient demographics across EMR, CRM, and scheduling tools.
3. Prioritize integration domains and use cases
You do not have to integrate everything at once. Sequence your work to address the highest impact domains first. For many health systems, these include:
• Patient and provider master data
• Encounters and episodes of care
• Orders and results for key specialties
• Claims and payer data for value‑based contracts
Align each domain with one or two priority use cases. For example, longitudinal patient view for high‑risk patients, or integrated claims and clinical data for risk adjustment analytics.
4. Decide on your integration pattern
For large health systems, point‑to‑point interfaces no longer scale. You need a pattern that standardizes how systems exchange data and how your teams manage change.
Common patterns include:
• API‑led integration using FHIR and REST APIs as the primary interface for new systems.
• Enterprise integration platform that centralizes message routing, transformation, and monitoring.
• Event‑driven architecture where source systems publish standardized events, such as patient updated or order created.
• Hybrid approach that supports HL7 v2, FHIR, flat files, and streaming data under a single governance model.
The goal is consistent hospital interoperability across new and legacy systems, not perfection in a single standard.
5. Standardize on data models and vocabularies
Agree on common definitions for core entities and fields:
• What defines a unique patient
• How you represent encounters and episodes
• Standard code sets for labs, diagnoses, procedures, and medications
Use industry standards where possible, including FHIR resources, SNOMED CT, LOINC, ICD‑10, and RxNorm. Apply these standards in your integration layer so that downstream systems receive consistent data, even if sources differ.
6. Build a master patient index and reference data layer
A strong master patient index reduces duplicate records and improves match quality across systems. It also supports safer EMR data consolidation and analytics.
In parallel, centralize reference data such as provider IDs, location IDs, and departments. Expose these reference data services to both operational systems and analytics environments. This step creates a shared source of truth that cuts across historical silos.
7. Integrate EMR data with ancillary and external sources
EMR data sits at the center of many workflows, but it is not the only critical source. You should integrate:
• Labs, radiology, cardiology, and pathology systems
• Population health tools and care management platforms
• Claims and payer portals
• Patient engagement and remote monitoring solutions
Use your integration layer to normalize and route data between these systems and your central repositories. For example, ingest external claims data and tie it to EMR encounters through your reference data and master patient index.
8. Operationalize interoperability in clinical workflows
Health system integration succeeds only when clinicians feel the difference. Focus on:
• Embedding external data views directly in the EMR
• Reducing clicks to see history from external providers
• Presenting reconciled medication and allergy lists
• Triggering alerts when critical data arrives from outside sources
Involve clinical leaders early. Test new views and workflows with pilot sites before wider rollout. Measure adoption and satisfaction, not only technical uptime.
9. Build a unified analytics layer on top of integrated data
Once you stabilize source integrations, focus on analytics. A unified data platform should:
• Ingest curated data from the integration layer
• Apply consistent business rules and metrics
• Support both governed reporting and advanced analytics
Your analytics environment should not re‑create healthcare data silos. Design it to support cross‑domain analysis and self‑service while preserving quality and security.
Governance Models
Data governance tied to clinical and operational outcomes
Data governance should not sit apart from daily work. Form a governance council that includes clinical, operational, IT, and analytics leaders. Assign each member responsibility for specific domains, such as patient, provider, encounters, or quality measures.
For each domain, define:
• Standard definitions and data owners
• Quality thresholds and validation rules
• Change management processes for new fields or code sets
• Access policies by role and purpose of use
Link these governance decisions to your core outcomes. For example, medication data standards tied to medication reconciliation quality metrics.
Integration governance and design authority
Establish an integration review board that approves how new systems connect to your environment. This group should:
• Validate that new vendor selections align with your integration standards
• Mandate use of your integration platform and patterns
• Review interface and API designs for reuse and consistency
• Track technical debt from exceptions and schedule remediation
This governance model helps you avoid new healthcare data silos created by unreviewed point solutions.
Security, privacy, and risk management
Security and privacy teams must sit inside your data and integration governance effort. Together, agree on:
• Data classification levels and minimum controls for each level
• Standardized logging and monitoring for all interfaces and APIs
• Procedures for third‑party risk assessment and ongoing review
• Protocols for incident response across connected systems
A joint model keeps security from becoming a late‑stage blocker. It also ensures that every new integration strengthens your overall risk posture rather than weakening it.
Lifecycle management and continuous improvement
Healthcare data strategy is not static. As your health system adopts new care models and digital tools, your integration priorities will shift.
Build a repeatable lifecycle:
• Review integration performance and adoption metrics quarterly
• Retire legacy interfaces and feeds when better options exist
• Evaluate new standards such as FHIR expansions and national networks
• Feed lessons from incidents and outages into new design guidelines
Continuous adjustment keeps your hospital interoperability program aligned with strategy instead of legacy constraints.
Conclusion
Healthcare data silos formed over years of growth, acquisition, and tactical decisions. They will not disappear with a single project or vendor. You need a clear strategy, practical sequencing, and governance that connects technology choices to clinical and financial outcomes.
By treating integration as a disciplined program, you can:
• Give clinicians a clearer, more complete patient picture
• Support value‑based care with reliable data across EMR and claims
• Reduce security and privacy risk from unmanaged data stores
• Free analytics teams to focus on insight instead of extraction
Vorro helps large health systems break down silos without ripping and replacing core systems. Vorro’s integration platform connects EMRs, ancillary systems, payers, and digital health tools through a flexible, standards‑aligned architecture. You gain consistent data flows, central monitoring, and a trusted foundation for your analytics and digital initiatives.
If you are ready to move from fragmented interfaces to strategic integration, connect with Vorro and design an integration roadmap that fits your health system.
FAQs
1. What are healthcare data silos in large health systems?
Healthcare data silos are isolated pockets of clinical or operational data that do not flow easily across systems or departments. Examples include separate EMRs for different hospitals, unintegrated specialty systems, local registries, and spreadsheets. These silos prevent clinicians, operations leaders, and analysts from seeing a complete, trusted picture of patients and performance.
2. How does health system integration improve patient outcomes?
Health system integration improves outcomes by giving care teams faster access to complete information. When data from EMRs, labs, imaging, and external providers flows into a unified view, clinicians have better context at the point of care. Integrated data also supports care coordination, risk stratification, and quality reporting, which strengthens value‑based care models and population health programs.
3. What is the role of EMR data consolidation in this effort?
EMR data consolidation reduces duplication and inconsistency across multiple EMR instances or vendors. It can involve migrating to a single EMR, normalizing data into a central repository, or both. The goal is a consistent source of truth for key patient and encounter data, available for both clinical workflows and analytics. Strong integration and master data management are essential to keep consolidated EMR data accurate over time.
4. How should I approach hospital interoperability with legacy systems?
Start by defining standards and integration patterns that future systems must follow, such as FHIR APIs and a central integration platform. For legacy systems, use adapters and integration tools that translate HL7 v2 and proprietary formats into your chosen standards. Prioritize high‑value use cases first, such as sharing medication history or test results, and then expand. Over time, you can retire the most limiting systems or reduce their scope.
5. What makes a healthcare data strategy effective?
An effective healthcare data strategy ties every integration and analytics effort to clear organizational outcomes. It defines target data domains, integration patterns, governance structures, and success metrics. It also addresses talent, operating model, and technology together. The result is a roadmap where each project moves you closer to safer care, better experiences, and stronger financial performance, instead of creating new silos.
